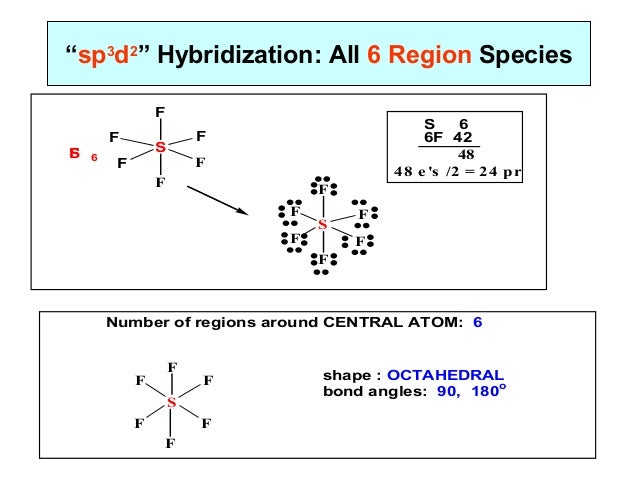
Linear: The sp hybridization is caused by the interaction of two-electron groups the orbital angle is 180°.All beryllium compounds, such as BeF 2, BeH 2, and BeCl 2, are examples.Each sp hybridised orbital contains the same amount of s and p characters.It’s also known as diagonal hybridization.
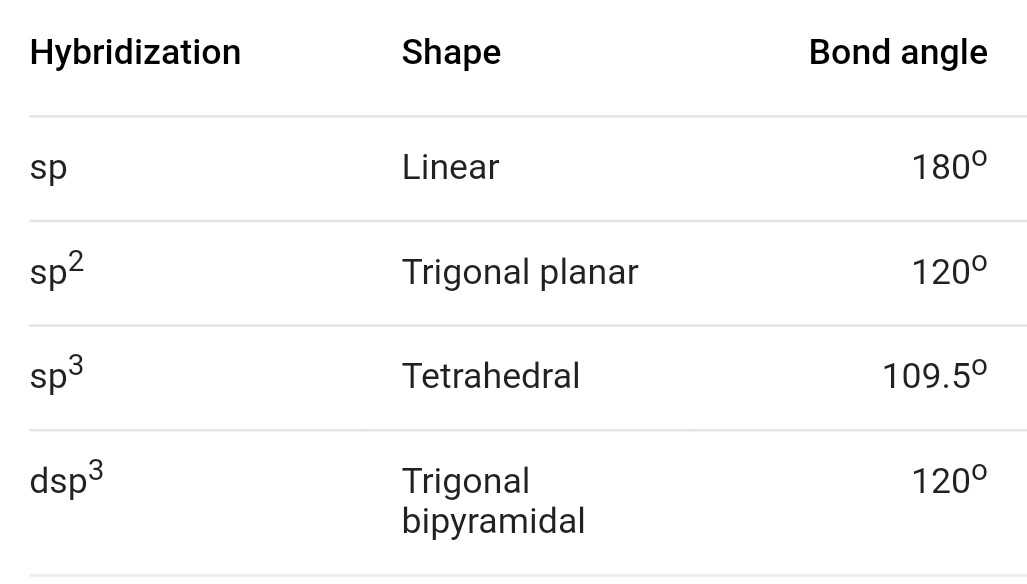
It entails combining one’s orbital and one ‘p’ orbital of equal energy to produce a new hybrid orbital known as an sp hybridised orbital. It produces linear molecules with a 180° angle. The newly formed orbitals are known as sp hybridised orbitals. It occurs when one s and one p orbital in an atom’s main shell combine to form two new equivalent orbitals. Hybridization can be classified as sp 3, sp 2, sp, sp 3d, sp 3d 2, or sp 3d 3 based on the types of orbitals involved in mixing. The larger lobe of the hybrid orbital is always positive, while the smaller lobe on the opposite side is always negative.If the hybridization of the molecule is known, the shape of the molecule can be predicted.Hybridization occurs only during bond formation, not in a single gaseous atom.Even orbitals that are completely filled but have slightly varying energy can participate. It is not required for all half-filled orbitals to participate in hybridization.The number of hybrid orbitals formed equals the number of atomic orbitals that mix.Hybridization occurs between atomic orbitals with equal energies.The atomic orbitals of comparable energies are mixed together during the hybridization process, which mostly involves the merging of two orbitals or two ‘p’ orbitals or the mixing of an ‘s’ orbital with a ‘p’ orbital as well as an ‘s’ orbital with a ‘d’ orbital. When two atomic orbitals combine to form a hybrid orbital in a molecule, the energy of the orbitals of individual atoms is redistributed to give orbitals of equivalent energy. The concept of hybridization is an extension of valence bond theory that helps us understand the bond formation, bond energies, and bond lengths. However, both fully filled and half-filled orbitals can participate in this process if their energies are equal. Hybridization is primarily carried out by atomic orbitals of the same energy level. This intermixing typically results in the formation of hybrid orbitals with completely different energies, shapes, and so on. The concept of hybridization is defined as the process of combining two atomic orbitals to create a new type of hybridized orbitals. Variation in Acceleration due to Gravity.Class 11 NCERT Solutions - Chapter 7 Permutations And Combinations - Exercise 7.1.General and Middle Terms - Binomial Theorem - Class 11 Maths.Average and Instantaneous Rate of Change.Class 11 NCERT Solutions - Chapter 3 Trigonometric Function - Exercise 3.1.Factors affecting Acceleration due to Gravity.What is the Difference between Interactive and Script Mode in Python Programming?.Difference Between Mean, Median, and Mode with Examples.ISRO CS Syllabus for Scientist/Engineer Exam.ISRO CS Original Papers and Official Keys.GATE CS Original Papers and Official Keys.
